Thermal Energy
This unit in our exploration of matter and energy delves into one of the more explosive forms of energy--heat. This corresponds to pages 255 - 271 in the textbook. This unit will involve cool things (like liquid nitrogen) and hot things like possibly rocketry.
What is heat really?
How is it measured?
How does it move around?
Heat is simply the total of the molecular movement in the object (in other words how much the atoms wiggle):

The faster the particles move, the hotter the object feels
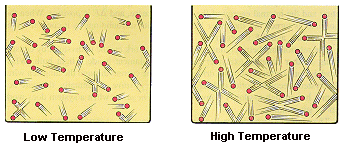


As you can see, with the same amount of molecules, the hotter a substance, the more space it takes up. Therefore, things expand when heated.
Hopefully you can also see that the more stuff in a space, the more heat it has (that is one of the reasons why it is colder on the top of a mountain).
 The more compressed the air, the more energy dense it is.
The more compressed the air, the more energy dense it is.
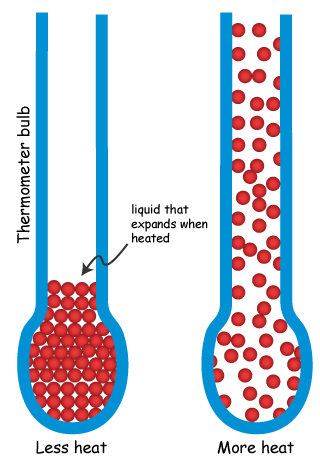
We use this expansion to help measure temperature (as well as propel rockets among other things).
There are other forms of thermometers, like this:
Inside them they have a strip with two different metals. Since the two metals expand and contract at different rates, heating the strip results in it bending:
They make sensitive thermometers out of balloons.
They also make thermometers that detect the infrared radiation given off by something (the sort that the doctor sticks in your ear and reads immediately).
Once you have something that changes with the temperature, you need to assign a scale to it.
There are three scales commonly used for measuring temperature:
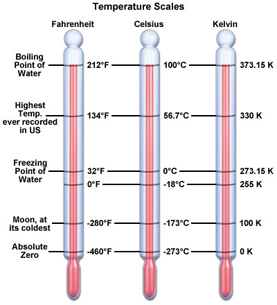
Fahrenheit was named by Daniel Fahrenheit in 1724. He set 0 at the temp he could cool brine and 100 at (roughly) human's temperature.
Anders Celsius set his scale by the freezing (0) and boiling (100) of water. Sometimes we call this scale "centigrade".
Lord Kelvin set his scale with 0 at the temperature where all movement stops--absolute zero--and kept the value of Celsius for each degree.

Heat is the measure of movement, so it does not sit still...it moves:
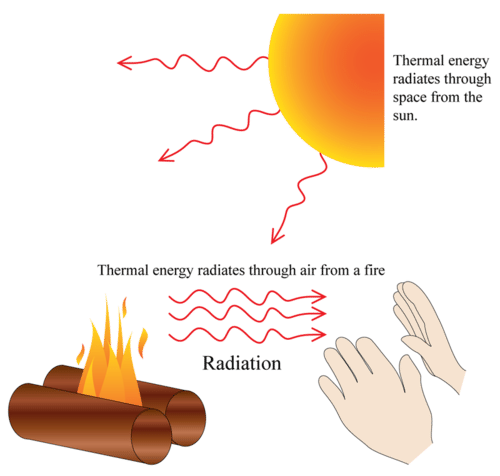
The three ways heat moves are conduction, convection, and radiation.
Conduction requires a solid (preferably metal). The molecules bump into each other and pass the energy along.
Things that allow heat to move well this way are called conductors. If they don't, they are called insulators.
What are good conductors? Insulators?


Convection requires a fluid. The heat warms the fluid which then rises and spreads.
This makes a convection current:
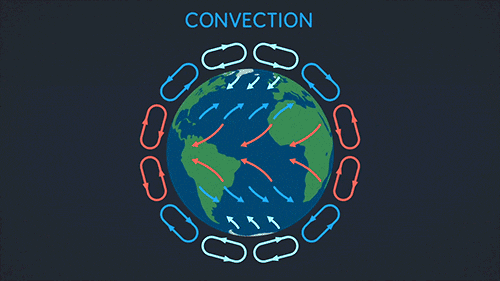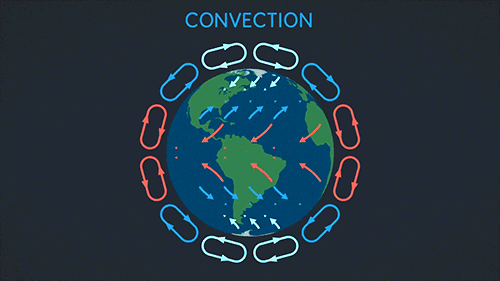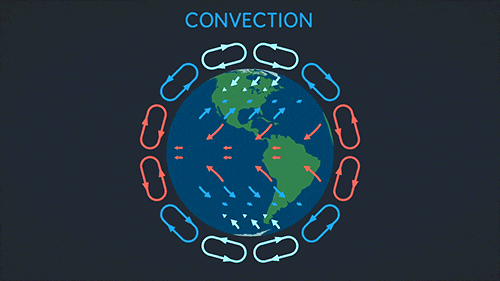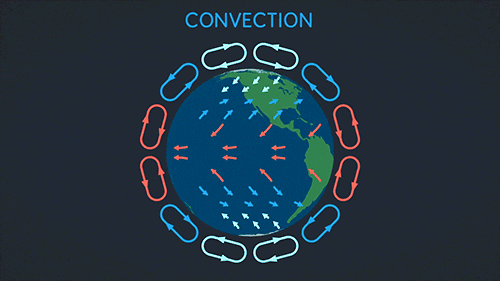
Radiation can go through a vacuum. Radiation is when heat energy moves through waves like sun light.
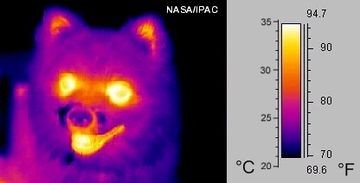
Not everything absorbs this radiation equally. What are the factors influencing radiation absorption? This will be a fun investigation. On a sunny day (or using a strong light inside), and a thermometer. See what heats up faster. Compare various things. Does color matter? How about texture? Shininess? Surface area? Size?
Some of the well known factors influencing radiation absorption are color, texture, surface area, sheen, and type of material.
This unit also deals with home heating systems. We would have compared hot water (the kind with a radiator) with hot air (the kind with vents). Which kind do you have at home?
Important things to understand:
What heat is.
Why do hot things expand?
Why is it cold on a mountain top?
How heat is measured?
What are the three scales commonly used to measure heat?
What is absolute zero?
What are the ways heat can be transferred.
What is a good conductor? Insulator?
Draw a convection current.
What are the factors influencing radiation absorption?